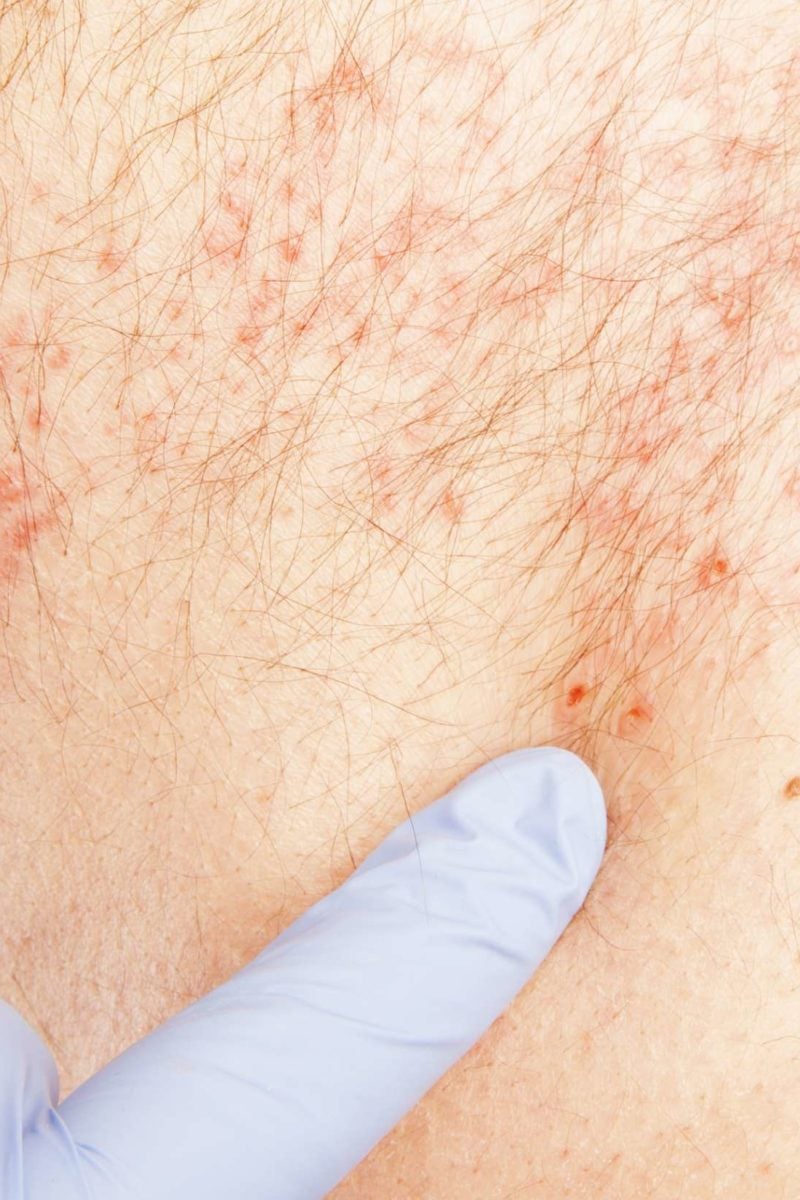
The report does not highlight any specific side effects which stood out among the reports of serious adverse events. 
The most common adverse events were fever, injection site pain, and injection site erythema. The MMWR findings come from data lifted from the Vaccine Adverse Event Reporting System (VAERS) during the first eight months Shingrix was in use, from Octoto June 30, 2018, resulting in the distribution of 3.2 million doses. It was the second shingles vaccine approved in the U.S., and has been widely viewed as a superior replacement for Merck’s Zostavax (zoster vaccine live ZVL), which has been linked to complications as a result of an “under-attenuated” live virus contained in the vaccine. Shingrix (recombinant zoster vaccine RZV) was introduced in October 2017, and has been approved for prevention of shingles among adults age 50 and older. However, it is unclear whether the deaths were tied directly to the vaccine. Centers for Disease Control and Prevention (CDC) Morbidity and Mortality Weekly Report (MMWR), health officials indicate that at least 4,381 reports of adverse events have been received involving shingles inoculation with Shingrix, about 130 of them are classified as serious, including seven deaths. GlaxoSmithKline’s Shingrix vaccine is believed to be safer and more effective for prevention of shingles among older adults, and has largely replaced the older live-virus Zostavax vaccine, which has been linked to reports of more severe and persistent shingles outbreaks and auto-immune complications in recent years. 
Amid continuing concerns about reports of problems with the single-dose Zostavax shingles vaccine, a new report reviewed adverse reactions to the newer, double-dose Shingrix vaccine, indicating that about three percent of those reactions resulted in serious injuries or complications.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
